Protoplast fusion
and genetic analysis
in Cephalosporium acremonium
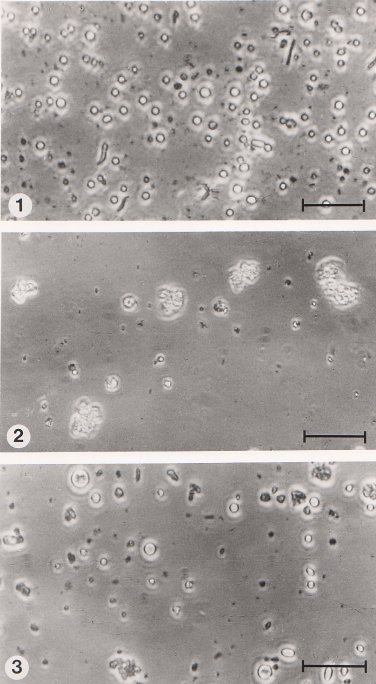
5.2.4 Fusion of protoplasts Protoplasts isolated from mutant strains were fused using polyethylene glycol (PEG), mol. wt. 6000 (Sigma), by a similar method to that described by Anné and Peberdy (1976) and Ferenczy et al. (1975). The protoplasts were separated from mycelial debris by slow centrifugation and washed twice in 0.7M NaCl. An equal number of protoplasts of each strain (108 of each) were mixed and centrifuged (700 g for 10 min). The pellet was then resuspended in 2 ml of 30% (w/v) PEG containing 0.01M CaCl2 and 0.05M glycine (adjusted to pH 7.5 with NaOH and sterilised by membrane filtration) and incubated for 10 min at 28oC. Finally, the suspension was diluted with 8 ml of 0.7M NaCl and centrifuged (700 g for 10 min). Treated protoplasts were washed twice with 0.7M NaCl and then resuspended in 10 ml of 0.7M NaCl. The overall process involves the aggregation of protoplasts in the presence of PEG as illustrated in Fig.5.4 and it is generally assumed that fusion occurs after the PEG has been diluted or washed away (Peberdy, 1980).
References Anné, J. and Peberdy, J. F. (1976). Induced fusion of fungal protoplasts following treatment with polyethylene glycol. Journal of General Microbiology, 92, 413-417. Ferenczy, L., Kevei, F. and Szegedi, M. (1975). High-frequency fusion of fungal protoplasts. Experientia, 31, 1028-1030. Peberdy, J. F. (1980). Protoplast fusion - a tool for genetic manipulation and breeding in industrial microorganisms. Enzyme and Microbial Technology, 2, 23-29. |

Figure 5.4 Aggregation and fusion of protoplasts induced by polyethylene glycol (PEG) and Ca2+ ions. (1) Suspension of freshly isolated protoplasts in 0.7 M NaCl. (2) Aggregation of protoplasts in the presence of PEG and Ca2+ ions. (3) Suspension of treated protoplasts in 0.7 M NaCl following the removal of PEG. Note the larger body probably a consequence of fusion between 2 or more protoplasts. The bar markers represent 30 µm. |
Copyright © 1982 Paul F Hamlyn
(http://fungus.org.uk/cv/thesis_fig5.4.htm)